J.S. Held Acquires Clark Seif Clark, Strengthening West Coast Capabilities for Environmental Claims, Disputes, and Catastrophe Response
Read MoreSpectroscopy is the study of the interaction of light and matter. In infrared spectroscopy, infrared light, which is electromagnetic radiation (EMR) with wavelengths longer than those of visible light, is shone either on or through material. The interaction between infrared light and the material creates a signal that can be used to understand its chemistry. Specialized infrared spectrographic instruments have been developed, the most useful of which is the Fourier Transform Infrared (FTIR) spectrometer. The industry uses three different types of FTIR spectrometers: Microscope FTIR, Bench FTIR, and a portable hand-held FTIR. The use of FTIR is typically nondestructive to the sample, so evidence is left intact and unaffected.
This paper will cover the basics of infrared spectroscopy and illustrate its usefulness in failure analysis investigations with several case studies. The following information may be of particular interest to insurance companies, litigants, manufacturers of plastic materials, forensic investigators, and anyone interested in understanding why materials fail.
FTIR is a spectroscopic technique used in the characterization and evaluation of materials by identifying the “chemical family” of a substance. When a material sample is analyzed, it produces a spectrum with absorption peaks corresponding to vibrational frequencies of bonds between the atoms making up the material. This infrared spectra functions like a chemical “fingerprint” which can be compared to reference databases to determine the chemical family or actual identity of unknown components and compounds. FTIR is also useful for identification of contaminants and chemical agents in contact with failed parts, and chemical bonds and sequences resulting from degradation such as from heat, water, oxygen, oxidizers, or ultraviolet (“UV”) light.
Applications of FTIR in failure analysis investigations include:
A manufacturer of disposable plastic baby bottle liners began receiving reports from consumers of contamination on the liners. Mold was initially suspected but ruled out after mycological analysis. Samples of customer returns and unopened boxes of production liners were obtained for analysis. An initial inspection showed two different types of contamination: light colored deposits as shown in Figure 1 and dark spots as shown in Figure 2.
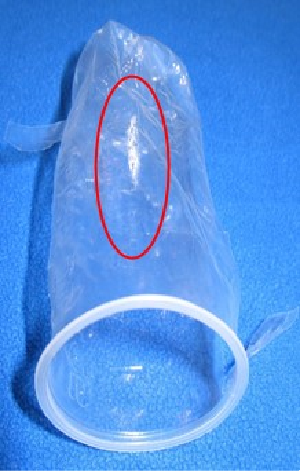

A small fragment (5/1000 inch) of the lighter colored contaminant, shown in the microscope view in Figure 3, was analyzed by FTIR and identified as containing phenyl phosphite (Figure 4), an anti-oxidant commonly used in processing of polyethylene (PE) to prevent thermal degradation at high temperatures. The FTIR spectra also contained extra peaks not associated with the antioxidant as shown by the red markings in Figure 4.

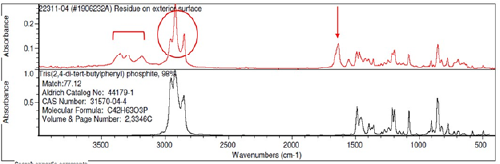
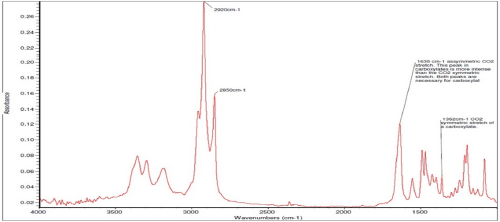
Figure 5 shows the result of spectral subtraction performed on the spectra to remove or “subtract” the phenyl phosphite peaks and reveal the masked or hidden substance(s). Peaks in the “fingerprint” region show strong evidence of a carboxylate such as a stearate. Stearates are wax-like or soap-like substances derived from fatty acids in vegetable oils or fats such as coconut and palm oils. Carboxylates can be identified by their characteristic peaks at wavenumber 1635 cm-1 and 1362 cm-1 with the former relatively more intense than the latter as evident in the FTIR spectra in Figure 5.
In plastics manufacturing, sheets of plastic or, in this case, the plastic liners, can stick together during processing. In order to mitigate this condition, additives, called slip or anti-block agents, are often used to lower the coefficient of friction so the plastic parts are able to slip past one another more easily. Erucamide, a waxy substance derived from rapeseed oil, and/or fine clays or talc are incorporated into the plastic and act as lubricants or anti-block agents. Only the additive that is present at the surface of the plastic sheet is effective as a slip or anti-block agent. Much of the clays and talc remain buried in the bulk of the plastic and are ineffective, whereas erucamide will “bloom” lightly to the surface but the concentrations will be so low as to not be visible. However, when excessive amounts of processing aids such as lubricants and slip compounds are used and/or a higher density resin is used than was intended (e.g., HDPE instead of LDPE), the additives can form deposits on surfaces of molds, dies, or other processing equipment and later slough off onto the plastic during production. The phenomenon is known as “plate-out” or “die drool.” Figure 6 shows an extreme example of the buildup on a plastic sheet extrusion machinery die. The analysis concluded that erucamide/anti-oxidant plate-out residue had accumulated on the die and deposited on the thin sheets that were later thermoformed into the baby bottle liners.

The black spots shown previously in Figure 2 were examined under a dissecting microscopic and determined to be embedded within the polyethylene liners and not on the surface of the material. Figure 7 shows one of the embedded black spots in cross-section after it was transected with a scalpel.

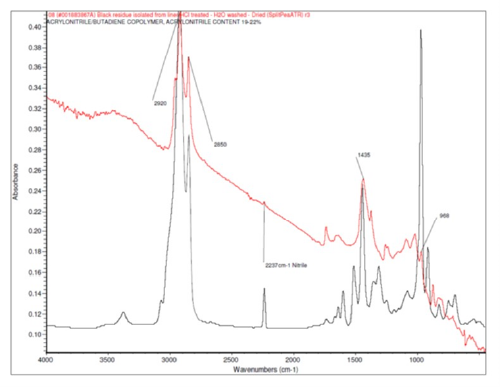
The black particles had a rubber-like consistency. FTIR analysis of the black spots did not positively identify the rubber-like material as it was masked by the numerous crystalline particles which overwhelmed the signal. The spectra indicated that the crystalls were calcium carbonate, a common filler used in rubbers to improve their physical properties. FTIR spectral subtraction was attempted as before but the results were inconclusive. A small amount of hydrochloric acid (HCl) was added to one of the particles to remove the calcium carbonate. HCl will dissolve calcium carbonate resulting in carbon dioxide gas and a water soluble salt that can be rinsed away leaving the rubbery material in isolation. Figure 8 shows digital microscope images of one black particle before and after the HCl treatment and rinsing. Note the large number of crystals in the as-is or pre-treatment image (left) and the relative lack of crystals in the post-treatment image (right).

Figure 9 is the FTIR spectra of the cleaned rubber particle and the spectra of Buna-N rubber showing an excellent match. Calcium carbonate filled Buna-N rubber is commonly used for rubber O-rings, washers, and gaskets. The contaminants were traced back to manufacturing equipment. The O-rings were found to have been deteriorating, resulting in small particles breaking off into the molten polyethylene and becoming embedded in the liners.
Reports described scuba air hoses failing internally and plugging up regulators that deliver breathing air to the diver. The failed hoses ranged in age from about 3-8 years and were generally located in tropical areas. The braided hoses showed no outward sign of deterioration; however, when the hoses were cut open, deterioration to the internal rubber hoses was clearly visible as shown in Figure 10 and Figure 11.


Subject failed hoses, new “brand-name” hoses, and “knock-off” brand hoses were obtained for examination and testing. Accelerated aging tests were performed to evaluate environmental effects on the deterioration of the hose materials. The new and knock-off rubber hoses were subjected to either dry air, hot water immersion, or 100% relative humidity conditions at 120oF. The knock-off hoses showed extensive signs of degradation similar to that of the subject failed hoses under wet and humid conditions, while the new brand-name hoses remained flexible and did not exhibit visible deterioration as shown in Figure 12.

Chemical FTIR analysis of the failed, knock-off, and brand name hoses showed that the hoses were made of thermoplastic polyurethane (TPU), a type of synthetic rubber. On a molecular level, the chemistry of TPU is referred to as a block copolymer that consists of alternating sequences of hard and soft segments which give TPU its rubber-like properties. These hard and soft segments can be chemically tied together with an “ether” or an “ester” linkage that give the TPU differing properties. The failed and knock-off hoses had similar chemical signatures; however, as shown in Figure 13, the spectra of the brand name hoses showed an additional peak at 1108 cm-1 Figure 13. The presence of this peak indicated that the brand name TPU contained a chemical bond called an “ether” linkage, and the lack of such a peak in the failed and knock-off TPUs indicated an “ester” linkage connecting the polyurethane portions of the molecule.
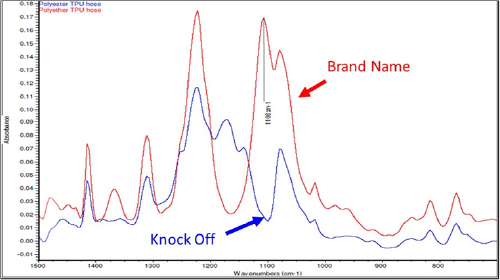
This distinction is important because TPUs with ester linkages (polyester TPU) are known to be susceptible to hydrolytic degradation while TPU with ether linkages (polyether TPU) are much more resilient to this type of decay. Testing and analysis showed that the combination of moisture in contact with the rubber hoses, the higher temperatures found in tropical environments, and, critically, the use of polyester (versus polyether) type TPU material for the hoses caused the rapid deterioration and failure of the scuba air hoses. Even though both air tubes were made from TPU, the specific polymer chemistry chosen to make the failed tubes was inappropriate for the application, being highly susceptible to environmental degradation and potentially leading to clogging of the second stage regulator of a scuba diver, in turn leading to loss of air supply. Consequently, the Consumer Products Safety Commission (CPSC) was notified, and corrective measures have been taken to address the issue.
A large, 400-foot-long hopper dredge ship, similar to the one pictured in Figure 14 operating in the Gulf of Mexico, was powered by three diesel engines that experienced a series of fuel injector failures after receiving fuel. The ship’s log showed the vessel lost approximately 22 injectors and, at one point, all propulsion was temporarily lost as the vessel was coming into port, which could have resulted in a collision with the dock or another vessel had one of the engines not restarted. The ship became so debilitated that it had to be taken out of service to affect repairs.

Laboratory analyses were conducted by third‐party labs on fuel samples from the refueling barge and debilitated dredge vessel. The problem was perplexing because the fuel injector failures occurred soon after refueling, and yet nothing from the fuel analysis explained these occurrences.
Incident fuel filter and fuel injectors were obtained for examination (See Figure 15 and Figure 16). Disassembly of the injector proved difficult as the needle valves were stuck in their housings. Once taken apart, wax‐like deposits were observed on the solenoid piston (see Figure 17) and light‐colored deposits were observed on the surfaces of the solenoids, needle valves, and needle valve housing bores as shown in Figure 18 and Figure 19. FTIR and SEM-EDS analyses were performed to provide chemical structure information and elemental composition of the deposits.





FTIR spectrum of all deposits, including the surface of the fuel filter, showed similar chemical structures. Representative spectra (shown in Figure 20) indicated “fatty acid soaps.” Elemental analysis of the deposits showed the present of sodium and/or calcium. These are the main counter ions in the fatty acid soap detected by FTIR.
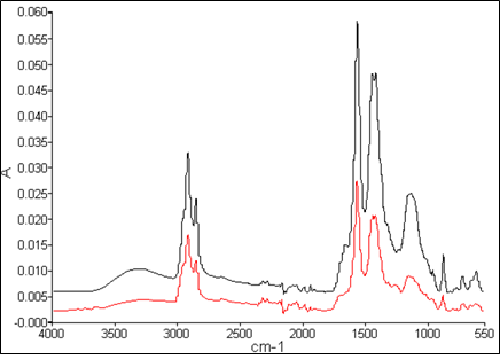
It was concluded that the soap deposits were likely responsible for the improper function of the injectors. Additional information regarding fuel processing and fuel additives led to the conclusion that the formation of the soap deposits was likely the result of two classes of contaminants in the fuel—acidic corrosion inhibitors; introduced during the refining, storage, and/or transport process prior to delivery to the vessel; and sodium and/or calcium. Possible sources of sodium and/or calcium include contamination from sea water, salt‐dryer filters used in the processing and refining of fuel, and chemicals used for cleaning the vessel’s fuel tanks. The third component that combined to form the soap deposits is the diesel fuel itself and/or biological growth (if water is present in the fuel). The soap formed was viscous and highly polar, capable of adhering to metal surfaces and “gumming up the works.” The presence of copper and zinc can accelerate such a process. Standard fuel tests currently in use were not effective for identifying the cause of the problem because the quantity of contaminants necessary for carboxylate formation which cause the injector failure was extremely small. The immediate solution to prevent injector failure was to prevent or remove formation of the deposits rather than try to identify the presence of contaminants in the fuel.
These findings were surprising because this was the first time the client was aware of failures of this sort having occurred in electronic unit injectors (EUI). Similar deposits have been known for some time to affect the trucking industry, where common‐rail fuel injection systems use higher‐pressure and closer‐tolerance injectors than EUIs. Because there are many older EUI‐equipped engines in service which are generally more tolerant of this problem than the common‐rail‐injected engines, such a discovery was concerning and may portend a spread to a larger hardware population.
Following the presentation of the findings, the diesel engine manufacturer began developing a test for the fuel that may reveal the presence of the reactants which led to the soap formation in the above scenario. Preliminary tests on a fuel sample from the vessel in question showed the presence of the reactants to be outside of the normal range, likely causing the formation of soap deposits. The engine manufacturer is continuing to refine test methods and hopes to soon have a commercially available field test.
Many different types of pipes are used in potable water service. Often, claims involve premature failure of polyethylene (PE) and cross-linked polyethylene (PEX) pipes used in household domestic water systems. Figure 21 shows a severely degraded PE water pipe in cross-section with cracks on the inner wall surface. FTIR can be a powerful tool in the analysis of PE and PEX when determining degradation.

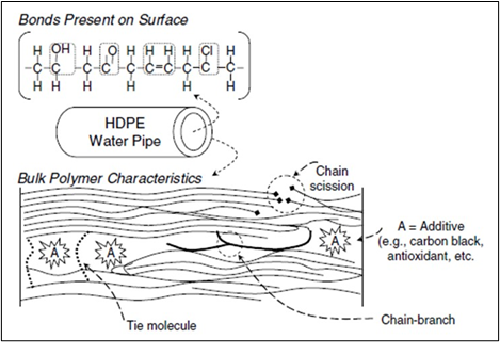
Most polymers, including plastic pipes, are compounded with antioxidants to prevent oxidation chain scission and the loss of molecular weight and mechanical properties. Antioxidants are sacrificial additives included in polymer formulations to counteract the oxidative effects of service environments. In water distribution pipes, the finite supply of antioxidants included in the pipe formulation is consumed by oxidation reactions. They can also be washed off the pipe surface by flowing water. The antioxidants are somewhat mobile and able to migrate within the pipe wall from the core to reaction regions on the inner diameter surface.
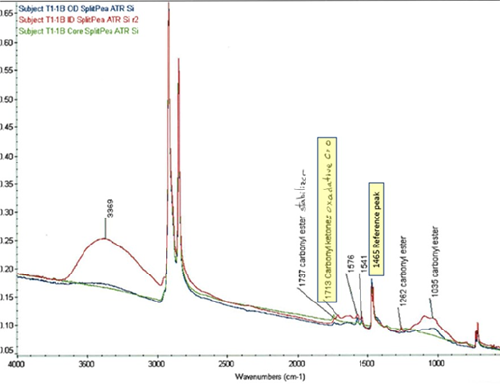
Service lifetimes for domestic water PE pipes is around 40 years. The working lifespan of pipes used in water distribution networks depends on the material’s retention of its antioxidant package. For PE pipes used in water distribution networks, the diffusion and loss of antioxidant from the material is driven from the inner pipe surface. When chlorine is present in the water, it can increase the depletion rate of the antioxidant. Figure 22 shows a representation of PE degradation in chlorinated drinking water systems. Oxidation of the PE leaves behind reaction species in the pipe wall such as vinyl groups (C=C), chlorine groups, and carbonyl groups (C=O). These species do not exist in significant amounts on non-degraded pipe. The presence of these chemical groups is one of the first indicators of degradation and can lead to polymer chain scission and mechanical failure resulting in crack propagation through the pipe wall. FTIR can detect these chemical bond changes that indicate the incipient oxidative processes before the pipe materials show visible signs of degradation. In particular, it is used to identify carbonyl groups that can be used to quantify the extent of oxidation through what has been termed the “Carbonyl Index” or CI. The carbonyl index can be calculated by taking a ratio of the height of the ketone C=O peak 1713 cm-1 to the height of reference peak of the backbone of the polymer 1465 cm-1 (Figure 23). Research has shown that carbonyl indices of the interior of polyethylene pipes below 0.02 indicate no significant oxidation while a CI above 0.1 denotes severe oxidization and embrittlement that may lead to pipe failure.
Infrared spectroscopy is highly useful in failure analysis and can be a great asset in cases in which it is critical to understand why and how a material failure occurred. Not only does FTIR allow for more efficient data collection, but it is also less invasive than traditional methods, provides greater spectral quality, and affords ease of use. Insurance companies, litigants, manufacturers of plastic materials, and others involved in matters wherein a material failure has occurred and given rise to relevant claims should consider involving spectroscopy experts as soon as possible in order to help ensure that proper and detailed materials analyses can be performed.
We thank our colleagues John Carroll, BS, Brian Flinn, PhD, PE, and Dale Clark, PE for providing insights and expertise that greatly assisted this research.
John Carroll is a Chemist in J.S. Held’s Forensic Architecture & Engineering Practice. John manages the chemistry laboratory at J.S. Held, Redmond and has more than 20 years’ experience in laboratory and field method development, research, and testing, using an extensive range of instrumentation. His project experience includes expert technical support and both development and utilization of chemical and instrumental analysis techniques. Capabilities at J.S. Held’s Material Science and Chemistry laboratory include analysis of polymers, fibers, and hydrocarbons by a variety of advanced techniques, such as infrared spectrometry (FTIR), differential scanning calorimetry (DSC), high-performance liquid chromatography (HPLC) as well as standard wet chemistry methodologies.
John can be reached at [email protected] or +1 425 207 4346.
Brian Flinn is a Vice President and Metallurgist in J.S. Held’s Forensic Architecture & Engineering Practice. His practice includes analysis of failure origins in metals, polymers/plastics/adhesives, composites, ceramics, glasses, concrete and wood products. Brian has been extensively involved in litigation matters involving personal injury, construction defects, corrosion and material degradation. He has also taken lead responsibility in pipeline leaks, medical implant failure cases, and patent infringement matters. Brian’s expertise with materials encompasses fatigue, fracture, and failure analysis, structure-property-processing relationships, interfacial structure and properties, adhesive bonding and joining, mechanical testing of materials, corrosion and degradation, and thermal analysis.
Brian can be reached at [email protected]
or +1 425 207 4358.
Infrared Thermography is the process of acquiring and analyzing thermal image information captured by a non-destructive thermal imaging device, often referred to as an infrared camera or an IR camera. These devices detect the heat...
The evaluation of complex losses often involves large volumes of information and the integration of numerous documents, which places added emphasis on comprehensive reporting that is easily accessible and understood. Used in conjunction with Building...